⊗This PRODUCT IS INTENDED AS A RESEARCH CHEMICAL ONLY. This designation allows the use of research chemicals strictly for in vitro testing and laboratory experimentation only. All product information available on this website is for educational purposes only. Bodily introduction of any kind into humans or animals is strictly prohibited by law. This product should only be handled by licensed, qualified professionals. This product is not a drug, food, or cosmetic and may not be misbranded, misused or mislabeled as a drug, food, or cosmetic.
⊗This PRODUCT IS INTENDED AS A RESEARCH CHEMICAL ONLY. This designation allows the use of research chemicals strictly for in vitro testing and laboratory experimentation only. All product information available on this website is for educational purposes only. Bodily introduction of any kind into humans or animals is strictly prohibited by law. This product should only be handled by licensed, qualified professionals. This product is not a drug, food, or cosmetic and may not be misbranded, misused or mislabeled as a drug, food, or cosmetic.
Glutathione 1500mg
Peptides
What is Glutathione?
Glutathione (L-γ-glutamyl-L-cysteinyl-glycine) is the most abundant non-protein thiol in mammalian cells, present in millimolar concentrations throughout the body with particularly high concentrations in the liver, kidneys, and immune tissues. This tripeptide serves as the master regulator of cellular redox homeostasis and functions as a critical cofactor in numerous enzymatic reactions essential for life.
The molecule exists in two primary forms: reduced glutathione (GSH), which constitutes more than 98% of total glutathione under normal physiological conditions, and oxidized glutathione (GSSG). The GSH:GSSG ratio serves as a sensitive marker of cellular oxidative status, with healthy cells maintaining ratios exceeding 100:1. This dynamic equilibrium is maintained by glutathione reductase, which regenerates GSH from GSSG using NADPH as a reducing agent.
Glutathione biosynthesis occurs exclusively in the cytosol through a two-step ATP-dependent process. The rate-limiting step involves glutamate-cysteine ligase catalyzing the formation of γ-glutamylcysteine from L-glutamate and L-cysteine, followed by glutathione synthetase adding glycine to form the complete tripeptide. Cysteine availability is the primary limiting factor in glutathione synthesis, making supplementation with cysteine precursors an effective strategy for increasing cellular glutathione levels.
The compound's unique γ-peptide bond between glutamate and cysteine renders it resistant to degradation by common peptidases, allowing intracellular accumulation to protective concentrations. Glutathione is compartmentalized throughout the cell, with 80-85% in the cytosol, 10-15% in mitochondria, and smaller amounts in the endoplasmic reticulum, nucleus, and peroxisomes. Mitochondrial glutathione is particularly critical for protecting against oxidative damage, as mitochondria lack the enzymes for glutathione synthesis and must import it from the cytosol via specific transport systems.
Glutathione levels naturally decline with aging, chronic disease states, and exposure to environmental toxins. This depletion compromises cellular defense mechanisms and contributes to numerous age-related pathologies including neurodegenerative diseases, cardiovascular dysfunction, metabolic disorders, and impaired immune function. The ability to maintain or restore optimal glutathione status represents a fundamental strategy for promoting healthspan and preventing oxidative stress-related pathological processes.
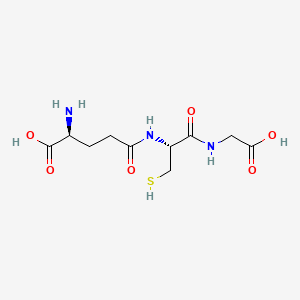
Glutathione Structure
Chemical Structure
2D Structure
3D Structure

Chemical Properties
| CAS Number | 70-18-8 |
|---|---|
| Molecular Formula | C10H17N3O6S |
| Molecular Weight | 307.33 g/mol |
| IUPAC Name |
(2S)-2-amino-5-[[(2R)-1-(carboxymethylamino)-1-oxo-3-sulfanylpropan-2-yl]amino]-5-oxopentanoic acid
|
| InChIKey | RWSXRVCMGQZWBV-WDSKDSINSA-N |
Glutathione Research
Exercise Performance and Muscle Recovery
Glutathione supplementation demonstrates significant effects on physical performance and exercise-induced fatigue. Research published in PMC shows that glutathione administration suppresses muscle fatigue induced by prolonged exercise through improved aerobic metabolism. In controlled studies, glutathione supplementation increased peroxisome proliferator-activated receptor-γ coactivator-1α (PGC-1α) protein levels by 25% and mitochondrial DNA biogenesis by 53% in skeletal muscle, indicating enhanced mitochondrial function supporting exercise capacity.
Human trials demonstrate that two weeks of glutathione supplementation (1g daily) significantly reduces exercise-induced blood lactate elevation and prevents the decrease in intermuscular pH that typically occurs during intense physical activity. Studies show glutathione-supplemented subjects experienced suppressed lactate accumulation during exercise, with placebo groups showing lactate levels of 3.4 mM compared to 2.9 mM in glutathione groups. Fatigue-related psychological factors measured by Profile of Mood State assessments were significantly decreased following glutathione supplementation.
N-acetylcysteine (NAC), a glutathione precursor, enhances performance in endurance athletes by increasing muscle glutathione availability. Research in the Journal of Applied Physiology demonstrates that NAC infusion improved time to fatigue at 92% VO2 peak by 26.3% in well-trained individuals. The mechanism involves NAC increasing muscle total glutathione by 25-40% and reduced glutathione levels in peripheral blood mononuclear cells by up to 100%, providing enhanced antioxidant capacity during intense exercise.
Animal studies show glutathione supplementation enhances post-exercise metabolic recovery by increasing PGC-1α expression and its downstream factors in skeletal muscle. The compound reduces plasma creatine kinase levels (a marker of muscle damage) and decreases malondialdehyde concentration (indicating reduced lipid peroxidation) in exercised muscle tissue. Glutathione modulates thioredoxin reductase activity in skeletal muscles, further supporting the antioxidant defense system during recovery periods.
Sources:
- Aoi W, Ogaya Y, et al. "Glutathione supplementation suppresses muscle fatigue induced by prolonged exercise via improved aerobic metabolism." Journal of the International Society of Sports Nutrition. 2015;12:7. https://pmc.ncbi.nlm.nih.gov/articles/PMC4328900/
- McKenna MJ, Medved I, et al. "N-acetylcysteine enhances muscle cysteine and glutathione availability and attenuates fatigue during prolonged exercise in endurance-trained individuals." Journal of Applied Physiology. 2006;100(5):1574-1582. https://journals.physiology.org/doi/full/10.1152/japplphysiol.00371.2004
- Watanabe N, Hoshi N, et al. "Oral administration of glutathione modulates exercise-related metabolic and oxidative factors in skeletal muscles of mice." Free Radical Research. 2024;58(1):62-73. https://www.sciencedirect.com/science/article/pii/S2667137924000110
Antioxidant Defense and Cellular Protection
Glutathione functions as the body's most powerful endogenous antioxidant system through multiple complementary mechanisms. Research published in Nature demonstrates that glutathione neutralizes reactive oxygen species (ROS) by donating electrons to quench free radicals, subsequently being oxidized to GSSG before regeneration by glutathione reductase. This redox cycling provides continuous protection against oxidative damage to proteins, lipids, and DNA.
The glutathione system operates through several enzymatic pathways that collectively protect against oxidative stress. Glutathione peroxidase, the most abundant peroxidase in cytosol and mitochondria, uses glutathione as a cofactor to convert hydrogen peroxide and lipid hydroperoxides into water and corresponding alcohols. Studies show glutathione's antioxidant capacity extends beyond direct ROS neutralization—it regenerates other oxidized antioxidants including vitamins C and E, creating a synergistic antioxidant network that amplifies cellular protection.
Research in Cell Death & Disease reveals that the glutathione system is essential for protecting cardiovascular tissue from oxidative injury. Studies demonstrate that glutathione restores mitochondrial respiration in type 2 diabetic heart tissue by improving mitochondrial bioenergetic function, increasing ATP production, and lowering oxidative stress levels. The compound's direct interaction with cellular proteins through S-glutathionylation provides protection against irreversible oxidation while serving as a redox signaling mechanism that modulates protein function.
Glutathione activates the Nrf2/HO-1 signaling pathway, a critical transcriptional regulator of antioxidant gene expression. Research shows glutathione supplementation significantly upregulates expression of antioxidant response element (ARE)-containing genes including heme oxygenase-1, superoxide dismutase, and catalase. This transcriptional activation provides sustained protection against oxidative stress by increasing cellular antioxidant enzyme capacity. Studies demonstrate that glutathione prevents hydrogen peroxide-induced apoptosis by rescuing mitochondrial membrane potential and blocking ROS accumulation, with protection dependent on HO-1 activation.
Sources:
- Aquilano K, Baldelli S, Ciriolo MR. "Glutathione: new roles in redox signaling for an old antioxidant." Frontiers in Pharmacology. 2014;5:196. https://www.frontiersin.org/journals/pharmacology/articles/10.3389/fphar.2014.00196/full
- Pizzino G, Irrera N, et al. "Protective Effect of Glutathione against Oxidative Stress-induced Cytotoxicity in RAW 264.7 Macrophages through Activating the Nuclear Factor Erythroid 2-Related Factor-2/Heme Oxygenase-1 Pathway." Antioxidants. 2019;8(4):91. https://pmc.ncbi.nlm.nih.gov/articles/PMC6523540/
- Xiao L, Xu X, et al. "Glutathione system enhancement for cardiac protection: pharmacological options against oxidative stress and ferroptosis." Cell Death & Disease. 2023;14:131. https://www.nature.com/articles/s41419-023-05645-y
Immune Function and Defense
Glutathione plays a fundamental role in regulating immune cell function and host defense mechanisms. Research in Proceedings of the Nutrition Society demonstrates that lymphocyte function is exquisitely sensitive to intracellular glutathione levels, with immune cells requiring a delicately balanced intermediate glutathione concentration for optimal performance. Studies show that interleukin-2-dependent functions including T-cell proliferation, CD8+ T-cell blast generation, cytotoxic T-cell activity, and natural killer cell function are particularly sensitive to glutathione depletion.
Controlled clinical trials demonstrate remarkable immune system enhancement following glutathione supplementation. Research published in the European Journal of Clinical Nutrition shows that oral liposomal glutathione administration (500-1000 mg daily) elevated natural killer cell cytotoxicity by up to 400% within two weeks and increased lymphocyte proliferation by 60%. At six months, glutathione levels increased 30-35% in erythrocytes, plasma, and lymphocytes, accompanied by significant reductions in oxidative stress markers including 35% decreases in plasma 8-isoprostane.
Studies in Immunity reveal that glutathione is essential for T cell metabolic reprogramming and effector functions. Research demonstrates that glutathione synthesis primes T cell metabolism for inflammatory responses through the AMPK-mTOR pathway, enabling proper glucose metabolism and cellular energetics required for immune activation. Glutathione restricts serine metabolism in regulatory T cells (Tregs), maintaining their suppressive capacity and preventing autoimmune responses. Genetic ablation of glutathione synthesis in Tregs leads to severe multi-organ inflammation, demonstrating glutathione's critical role in immune homeostasis.
The compound enhances cytotoxic T lymphocyte responses and prevents immunological unresponsiveness. Research shows that glutathione administration at the time of immunization reconstitutes suppressed CTL responses when nonprofessional antigen-presenting cells are used at high doses. Professional antigen-presenting cells like macrophages release substantial amounts of cysteine, raising intracellular glutathione levels in activated T cells and supporting optimal immune function. In disease states associated with glutathione deficiency, including HIV infection, cysteine supplementation significantly increases immunological functions including near-complete restoration of natural killer cell activity.
Sources:
- Dröge W, Breitkreutz R. "Glutathione and immune function." Proceedings of the Nutrition Society. 2000;59:595-600. https://www.cambridge.org/core/journals/proceedings-of-the-nutrition-society/article/glutathione-and-immune-function/39488A395CB25E88283204A938A4CBD9
- Sinha R, Sinha I, et al. "Oral supplementation with liposomal glutathione elevates body stores of glutathione and markers of immune function." European Journal of Clinical Nutrition. 2018;72:105-111. https://www.nature.com/articles/ejcn2017132
- Mak TW, Grusdat M, et al. "Glutathione Primes T Cell Metabolism for Inflammation." Immunity. 2017;46(4):675-689. https://pubmed.ncbi.nlm.nih.gov/28423341/
Aging, Longevity and Mitochondrial Function
Glutathione deficiency is a hallmark of aging that contributes to age-related metabolic decline and mitochondrial dysfunction. Research published in Innovation in Aging demonstrates that supplementation with glutathione precursors (N-acetylcysteine and glycine) prevents age-related decline and significantly increases longevity. In controlled studies using aging C57BL/6J mice, supplementation beginning at 65 weeks increased lifespan by 23.7% (104.0 vs. 128.6 weeks, p<0.0001), representing a substantial extension of both lifespan and healthspan.
The mechanism underlying glutathione's longevity effects involves restoration of mitochondrial fatty-acid oxidation capacity. Studies in 90-week-old mice show that eight weeks of glutathione precursor supplementation significantly improved glutathione status, reduced oxidative stress, enhanced mitochondrial fat-oxidation, and upregulated molecular regulation of mitochondrial energy metabolism. These findings demonstrate that preventing age-related glutathione deficiency preserves mitochondrial energetics critical for maintaining physiological function during aging.
Clinical research in older humans confirms these mechanisms translate to human aging. A randomized controlled trial published in PMC demonstrates that older humans (71.1 years) exhibited significantly lower glutathione levels, impaired mitochondrial fatty-acid oxidation, reduced gait speed and physical function, and higher oxidative stress compared to young controls (25.5 years). Sixteen weeks of GlyNAC supplementation in older adults significantly restored mitochondrial function, increased gait speed by 19%, improved physical function, and decreased oxidative stress (TBARS) by 80% and inflammation markers.
Research in GeroScience shows that mitochondrial glutathione plays a key role in protecting against age-associated oxidative damage. Aging is associated with decreased reduced glutathione in cells and increased oxidized glutathione, indicating a pro-oxidizing shift in redox balance. Mitochondrial glutathione depletion directly correlates with oxidative damage to mitochondrial DNA, which accumulates during aging and impairs respiratory chain function. Studies demonstrate that the ability of animal species to protect mitochondrial DNA through glutathione-dependent mechanisms is directly proportional to longevity across species.
Sources:
- Kumar P, Liu C, Sekhar R. "REVERSING AGING: PREVENTING AGE-RELATED DECLINE IN GLUTATHIONE AND MITOCHONDRIAL FUNCTION INCREASES LONGEVITY." Innovation in Aging. 2018;2(suppl_1):887. https://academic.oup.com/innovateage/article/2/suppl_1/887/5184000
- Kumar P, Osahon O, Sekhar RV. "CORRECTING GLUTATHIONE DEFICIENCY AND MITOCHONDRIAL DYSFUNCTION IN OLDER HUMANS: A RANDOMIZED CLINICAL TRIAL." The FASEB Journal. 2019;33(S1). https://pmc.ncbi.nlm.nih.gov/articles/PMC6840014/
- Hagen TM, Vinarsky V, et al. "Glutathione, oxidative stress and aging." GeroScience. 2002;24(1):4-9. https://link.springer.com/article/10.1007/BF02434082
- Sekhar RV, Patel SG, et al. "Deficient synthesis of glutathione underlies oxidative stress in aging and can be corrected by dietary cysteine and glycine supplementation." American Journal of Clinical Nutrition. 2011;94(3):847-853. https://pmc.ncbi.nlm.nih.gov/articles/PMC3155927/
Detoxification and Heavy Metal Chelation
Glutathione serves as the primary mechanism for Phase II hepatic detoxification, facilitating the elimination of xenobiotics, environmental toxins, and heavy metals from the body. Research demonstrates that glutathione is the most abundant biothiol in the liver at approximately 10 mM concentration, where it undergoes conjugation reactions with toxic compounds through glutathione S-transferase (GST) enzymes. This conjugation process transforms lipophilic toxins into water-soluble glutathione adducts that can be efficiently eliminated through bile or urine.
Studies published in PMC reveal that glutathione-mediated biotransformation is essential for eliminating heavy metals including cadmium, mercury, lead, and arsenic. The cysteine residue in glutathione possesses strong affinity for heavy metals through its sulfhydryl group, forming stable metal-glutathione complexes that prevent toxic metal accumulation in tissues. Research shows glutathione forms complexes with cadmium primarily through deprotonated sulfhydryl groups from cysteine residues, enabling chelation and sequestration of the toxic metal for safe elimination.
Animal studies demonstrate that heavy metal exposure significantly depletes hepatic glutathione levels while increasing GST enzyme activity as a compensatory response. Research shows that administration of cadmium chloride, mercuric chloride, lead acetate, and silver nitrate causes substantial glutathione depletion, with percentage depletion increasing further after repeated exposures. The resulting alterations in Phase II drug-metabolizing enzyme activities significantly affect hepatic capacity for detoxification of endogenous and exogenous toxic compounds.
Glutathione conjugation facilitates elimination of diverse environmental toxins including solvents, herbicides, fungicides, polycyclic aromatic hydrocarbons, and lipid peroxides. Studies show that glutathione conjugates bind to lipophilic xenobiotics and facilitate their excretion through specialized efflux transporters. This detoxification system becomes compromised when toxin exposure exceeds the body's capacity to produce or absorb glutathione, leading to toxin accumulation and cellular dysfunction. Research demonstrates that maintaining optimal glutathione levels is essential for protection against environmental toxin exposure and for supporting the body's continuous detoxification requirements.
Sources:
- Cobbina SJ, Chen Y, et al. "Glutathione Is a Key Player in Metal-Induced Oxidative Stress Defenses." International Journal of Molecular Sciences. 2012;13(3):3145-3170. https://pmc.ncbi.nlm.nih.gov/articles/PMC3317707/
- Sheweita SA, Tilmisany AK. "Heavy Metal-Induced Changes in the Glutathione Levels and Glutathione Reductase/Glutathione S-Transferase Activities in the Liver of Male Mice." Journal of Toxicology and Environmental Health. 1998;53(8):595-608. https://journals.sagepub.com/doi/10.1080/109158198226224
- Delalande O, Desvaux H, et al. "Cadmium–glutathione solution structures provide new insights into heavy metal detoxification." The FEBS Journal. 2010;277(24):5086-5096. https://febs.onlinelibrary.wiley.com/doi/10.1111/j.1742-4658.2010.07913.x
- He P, Lu X, et al. "Glutathione-mediated biotransformation in the liver modulates nanoparticle transport." Nature Communications. 2019;10:2647. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7252432/
Tissue Repair and Wound Healing
Glutathione plays multifaceted roles in wound healing through regulation of oxidative stress, collagen synthesis, and cellular proliferation. Research published in Plastic and Reconstructive Surgery demonstrates that topical glutathione treatment in ischemic wound models significantly reduces oxidative stress and reestablishes the matrix metalloproteinase-1/tissue inhibitor of metalloproteinase-1 (MMP-1/TIMP-1) ratio, enabling adequate extracellular matrix production and reepithelization. Studies show esterified glutathione effectively promotes healing in ischemic wounds where impaired blood flow compromises tissue repair.
The compound's effects on collagen deposition are mediated through complex interactions with transforming growth factor-beta (TGF-β) signaling. Research in Archives of Biochemistry and Biophysics shows that TGF-β decreases intracellular glutathione content in fibroblasts, triggering increased collagen I mRNA and protein production. Prevention of glutathione depletion with N-acetylcysteine or glutathione esters abrogates TGF-β-stimulated collagen production, while deliberate glutathione depletion enhances collagen synthesis. These findings indicate glutathione status regulates collagen production through modulation of reactive oxygen species signaling pathways essential for wound repair.
S-nitrosoglutathione (GSNO), a nitrosylated derivative, demonstrates enhanced wound healing properties. Studies show GSNO administration increases wound collagen content by 52.0% at day 5 and 47.5% at day 10 compared to controls, while maintaining physiological collagenolytic activity necessary for proper tissue remodeling. Research indicates GSNO increases systemic nitrite levels and delivers nitric oxide to wound sites, promoting enhanced fibroblast collagen synthesis without interfering with matrix metalloproteinase activity required for balanced extracellular matrix turnover.
Diabetic wound healing, characterized by delayed closure and impaired tissue repair, is significantly improved by glutathione-based interventions. Studies in diabetic animal models show that antioxidant treatment with N-acetyl cysteine and α-tocopherol dramatically reduces oxidative stress, increases antibiotic sensitivity of biofilms, and promotes proper collagen deposition and remodeling. Research demonstrates healing improved within 30 days following antioxidant treatment compared to 100 days for untreated controls. Investigations reveal that glutathione depletion in diabetic wounds contributes to delayed healing, and restoration of glutathione status through precursor supplementation supports normalized tissue repair processes.
Sources:
- Bozkurt A, Cakir B, et al. "Effects of topical glutathione treatment in rat ischemic wound model." Annals of Plastic Surgery. 2007;58(4):449-455. https://pubmed.ncbi.nlm.nih.gov/17413890/
- Liu RM, Gaston Pravia KA. "Glutathione regulates transforming growth factor-beta-stimulated collagen production in fibroblasts." Archives of Biochemistry and Biophysics. 2003;417(1):38-45. https://pubmed.ncbi.nlm.nih.gov/12959930/
- Schäffer MR, Tantry U, et al. "Nitrosoglutathione triggers collagen deposition in cutaneous wound repair." Plastic and Reconstructive Surgery. 2005;116(4):1035-1041. https://pubmed.ncbi.nlm.nih.gov/16008727/
- Maver T, Smrke DM, et al. "The Role of Antioxidants on Wound Healing: A Review of the Current Evidence." International Journal of Molecular Sciences. 2021;22(11):6084. https://pmc.ncbi.nlm.nih.gov/articles/PMC8397081/


